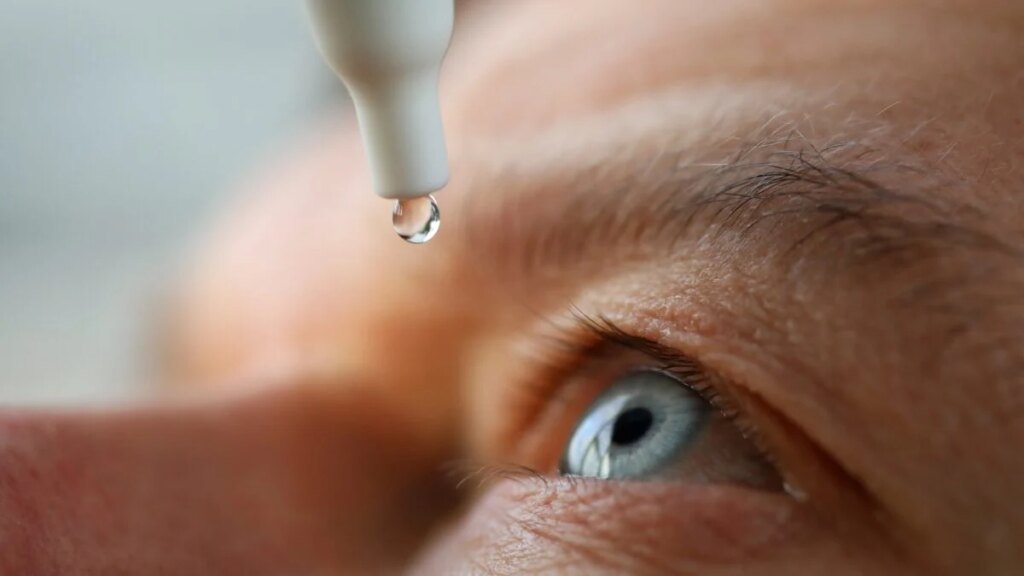
Over 3 million bottles of eye drops have been recalled over concerns about sterility, according to a notice at the FDA’s website. The eye drops have been sold under a variety of names at the most popular drug stores and supermarkets in the U.S.
The eye drops fall into eight main categories, manufactured by K.C. Pharmaceuticals in Pomona, California, though each category contains many different brands and store names.
The FDA says no press release has been issued about the recall and K.C. Pharmaceuticals didn’t respond Monday to emailed questions about what consumers should do with the recalled products. Gizmodo will update this article if we hear back from the company.
1) Sterile Eye Drops A.C.
- Best Choice, distributed by Valu Merchandisers
- QC – Quality Choice, distributed by CDMA, Inc.
- Discount Drug Mart
- Good Sense, distributed by Perrigo Direct
- Walgreens
- CAREONE, distributed by Foodhold U.S.A.
- Equaline, distributed by UNFI
- TopCare health, distributed by Topco Associates
- Meijer, distributed by Meijer Distribution
- H.E.B.A.C., made by H-E-B
2) Eye Drops Advanced Relief
- Walgreens
- Kroger
- CVS
- QC – Quality Choice, distributed by CDMA
- Discount Drug Mart
- Foster & Thrive, distributed by McKesson Corp.
- GoodSense, distributed by Perrigo Direct
- TopCare Health, distributed by Topco Associates
- DG Health, distributed by Old East Main Co.
- Fast Acting Code Red Eye Drops, distributed by 4 Trees
3) Dry Eye Relief Eye Drops
- GeriCare
- TopCare Health
- Rite Aid
- Leader, distributed by Cardinal Health
- Discount Drug Mart
- H‑E‑B
- Foster & Thrive
- Meijer
- DG Health
- Harris Teeter
- Exchange Select, manufactured for military exchanges
- Good Neighbor Pharmacy, distributed by Amerisource Bergen
4) Ultra Lubricating Eye Drops
- Leader, distributed by Cardinal Health
- Harris Teeter
- Foster & Thrive, distributed by McKesson Corp
- Walgreens
- P High Performance, distributed by Publix
- Kroger
- Good Sense Ultra, distributed by Perrigo
- Meijer
- CVS Health
- QC – Quality Choice, distributed by CDMA
- Avenova, distributed by NovaBay Pharmaceuticals
5) Sterile Eye Drops Original Formula
- Good Sense, distributed by Perrigo Direct
- Walgreens
- Kroger
- Lil’ Drug Store Products
- CVP, distributed by Salado Sales
- Harris Teeter
- H‑E‑B
- Rugby
- Equaline, distributed by UNFI
- Discount Drug Mart
- Leader, distributed by Cardinal Health
- CareOne, distributed by Foodhoold U.S.A.
- Circle K manufactured by Lil’ Drug Store Products
- DG Health, distributed by Old East Main Co.
- Good Neighbor Pharmacy, distributed by Amerisource Bergn
- TopCare Health, distributed by TopCo Associates
- Best Choice, distributed by Valu Merchadisers
6) Sterile Eye Drops Redness Lubricant
- Industrial Eye Relief, distributed by Cintas Corp.
- Good Neighbor Pharmacy, distributed by Amerisource Bergen
- Colirio Ojo de Aguila, distributed by Grandall Distributing Co.
- Colirio Oftal‑Mycin, distributed by Grandall Distributing Co.
- Leader, distributed by Cardinal Health
- Equaline, distributed by UNFI
- Walgreens
- Foster & Thrive
- Rite Aid
- Discount Drug Mart
7) Sterile Eye Drops Soothing Tears
8) Artificial Tears Sterile Lubricant Eye Drops
- Leader, distributed by Cardinal Health
- Good Sense, distributed by Perrigo Direct
- Good Neighbor Pharmacy, distributed by Amerisource Bergn
- TopCare Health, distributed by Topco Associates
- Best Choice, distributed by Valu Merchadisers
- Kroger
- P, distributed by Publix
- Quality Choice, distributed by CDMA
The FDA’s website has the complete list of UPC numbers for each brand name that has been recalled.
There were several eye drop recalls in 2023 when drug-resistant bacteria was discovered in many store brands. There were four deaths and at least 14 cases of vision loss traced to that contamination, according to the Associated Press. Four people had to have an eye removed. There are no known serious illnesses linked to this new recall.